AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Iron cathode reaction11/13/2023 Phenol Phthalein becomes to reddish pink colour while in the presence of OH –. 
The colour presence is used due to the two indicators in the test which is performance in lab includes: Copper becomes the cathode and the same decrease reaction of water and oxygen to OH‾ ions occur here( ).įigure 2 shows Galvanic corrosion of an iron nail half copper plated at the pointed end.įigure 3 illustrates which there is a combination of the dark bluish and reddish-pink colour. Iron is the more sensitive of the two metals thus it oxidation to generate Fe²+ ions producing the potassium ferracyanide become to blue. Copper is more noble than iron, so it turned to pink area or cathode and the iron turned to blue are or an anode. As the metals are straight interaction, corrosion happens. Picture two shows an iron nail half copper plated at the pointed end plunged in the electrolytic solution. The low corrosion resistant metal can be anodic, and destroy or corrode, when the rather corrosion resistant matters can be catholic, after that covered from corrosion (Fontana, 1978). They are electrically connected, electrons movement from one metal to another. When two unlike metals are submerged and plunged in a conductive environment where exist a potential difference among them. (Richard Schwartz, Chairman & Founder 1996) The major reddish colour area is the cathodic region, the minor quantity seen on the peak could display some reduction happening due to a dirty nail or any cold working included in production. There is shown a minor quantity of blue colour on the bend and in the area this could be due to inadequacies in the oxide substrate. This test contains a bent iron nail which is illustrated the anode by the blue colour potassium ferricyanide at the peak of the end. The chemical reaction in this illustration is:Īt the an anode (blue area): Fe → Fe2 + + 2e –Īt the a cathode (pink area): 2H 2O + O 2+ 4 e – –> 4OHįigure 1 shows iron bent nail, corrosion due to differential aeration 
The iron nail is contacted with a corrosive environment, with chemical condition within in order to display the presence of some kind of ions. The anode is illustrated in blue are or Fe2 + and the cathode is illustrated in red area or of OH –. The figure one shows an example of this practice happening in an iron nail. The cathode will be shaped where more oxygen is existing. The relative obtainability of oxygen in various sections of the metal surface is one of the things which influence the configuration of anodes and cathodes. The phenol phthalein acts red in the existence of OH – ions and the potassium ferri-cyanide acts blue in the existence of iron ions. Sodium chloride with agar gel operates as the electrolytic conductor, and the phenol phthalein solutions and potassium ferri-cyanide operates as chemical indicators. There were used many Petri dishes, large sufficient to accommodate the nails. there are used wires and clamps to connect the nails and zinc strips. There were used three iron nails, half copper plated at the pointed end, one iron nail half zinc plated at the pointed end, two strips of thin zinc and mild steel sheet were used submerged into agar gel including 3% sodium chloride, 5% potassium ferri-cyanide solution ,and 1% phenol phthalein solution. The tests have done in the lab illustration different kind of aqueous corrosion which is: There is needed an anode (Oxidation happens) ,and Cathode ( Reduction happens) The corrosion of different metals in an aqueous environment are an electrochemical specific action containing an electrolytic conductor, generally Acid + Alkali= Salt + water, the metal, an electronic conductor, the arrangement of cathode and anode, which can be dissimilar metals or dissimilar parts of metal.Ī chemical reaction occurs where oxidation and reduction happen at the same position. 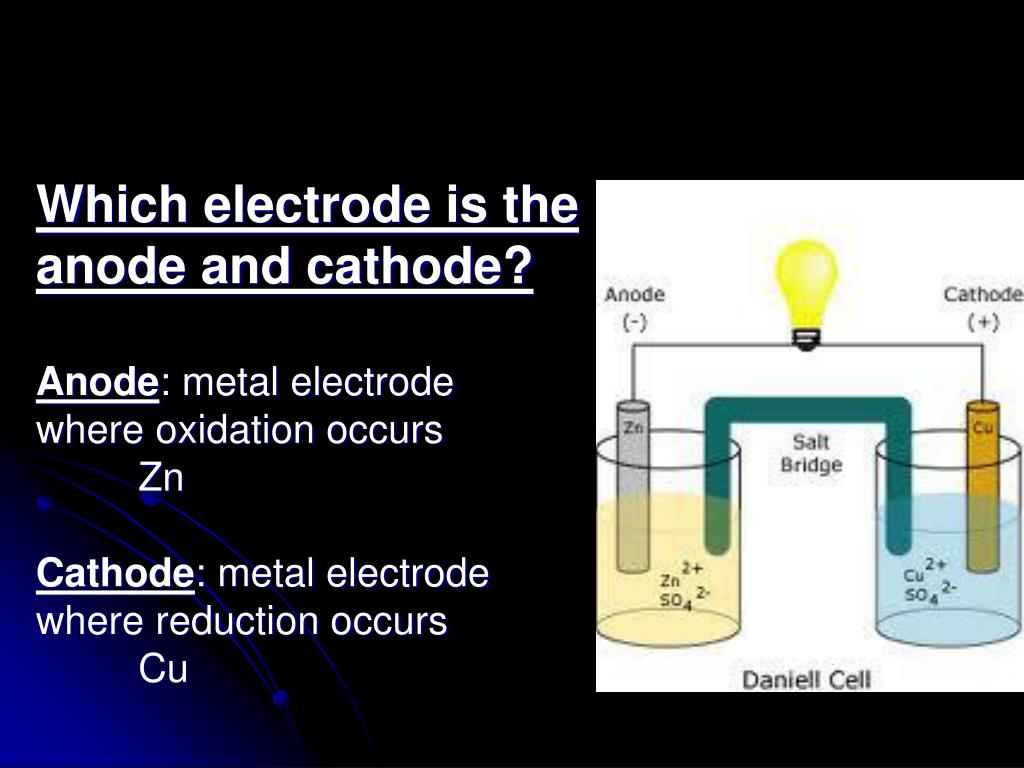
Galvanic corrosion and differential corrosion Laboratory report task 1 in sheaf building on Monday 20 October 2014
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
